Å@
Ichiro Fujimoto1, Kyoko Shirakabe1, Hidetoshi Miyashita1, Katsuhiko Mikoshiba1,2,
and Keiichi Torimitsu1,3
1Divisions of Neural Signal Information NTT-IMSUT and 2Molecular Neurobiology, The Institute of Medical Science, University of
Tokyo, 3Materials Science Laboratory
Å@The pH in the cell changes dynamically and the pH balance is regulated
by carbon dioxide in vivo. It is converted into bicarbonate and transported through the transmembrane,
and the cellular pH is adjusted. Bicarbonate is mainly secreted from gland
cells of pancreas and works as an acid neutralizer in the stomach. Kidney
cells control arterial blood pH. Several diseases or conditions such as,
glaucoma, cataract, cornea disease, stunted growth, and mental retardation
have been associated with the collapse of the pH balance.
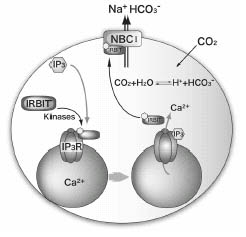
Å@Inositol 1,4,5-trisphosphate (IP3) receptors (IP3Rs) are IP3-gated Ca2+ channels located on intracellular Ca2+ stores. We previously identified an IP3R binding protein, termed IRBIT (IP3 Rbinding protein released with inositol 1,4,5-trisphosphate). Since IRBIT is released from IP3R by physiological concentrations of IP3, we hypothesized that IRBIT is a signaling molecule that is released from
IP3R and regulates downstream target molecules in response to the production
of IP3. We therefore attempted to identify the target molecules of IRBIT in this
study and succeeded in identifying Na+/HCO3- cotransporter 1, NBC1, as an IRBIT binding protein. Of the two major splicing
variants of NBC1, pancreas-type NBC1 (pNBC1) and kidney-type NBC1 (kNBC1),
IRBIT was found to specifically bind to pNBC1 and not to bind to kNBC1
at all. IRBIT binds to the N-terminal pNBC1-specific domain, and its binding
is dependent on the phosphorylation of multiple serine residues of IRBIT.
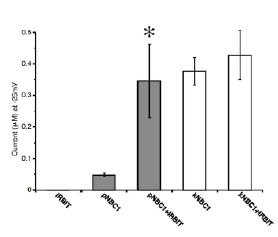
Å@Furthermore, an electrophysiological analysis in Xenopus oocytes revealed that pNBC1 requires co-expression of IRBIT to manifest
substantial activity comparable to that of kNBC1, which displays substantial
activity independently of IRBIT. These results strongly suggest that pNBC1
is the target molecule of IRBIT and that IRBIT plays an important role
in pH regulation through pNBC1.
Å@In addition, our findings raise the possibility that the regulation through IRBIT enables NBC1 variants to play different physiological roles.
[1] K. Shirakabe et al., Proc Natl Acad Sci USA 103 (2006) 9542-9547.
 |
 |
||||
|
|