Materials Science Laboratory
A lipid bilayer, which is a fundamental component of a cell membrane,
can be spread artificially on a substrate by self-assembly. Using this
self-spreading nature of an artificial cell membrane (ACM), we have investigated
the effect of a single nanogap structure on the molecular dynamics within
an ACM [1, 2]. We report here the electrostatic control of ACM self-spreading
by the temporal switching of an electric field applied to a nanogap structure
[3].
A pair of electrodes with a nanogap was fabricated using Au/Ti. A 10 µm
wide microchannel with wells at both ends was fabricated on this nanogap
structure. A lipid mixture consisting of Egg-PC and Egg-PG (molar ratio
7:3) containing 1 mol% Texas Red-DHPE was attached to one of the wells.
The self-spreading of an ACM was initiated by immersing the substrate in
a buffer solution containing 0.1-100 mM NaCl.
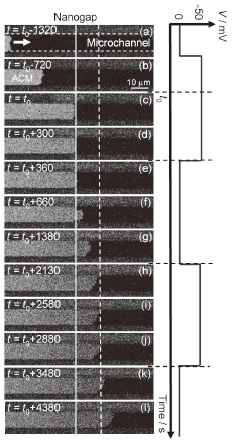
Figure 1 shows a typical time evolution for a self-spreading ACM induced
by the temporal switching of the applied voltage when we used a 100 mM
NaCl solution and a nanogap with a separation of <5 nm. Before the ACM
passed through the nanogap, no voltage-dependent changes in the self-spreading
were observed [Fig. 1(a) and (b)]. However, when the ACM reached the nanogap,
the self-spreading was forcibly prevented by the voltage application [Fig.
1(c)]. This continued for 〜300 s, corresponding to the interval of the
voltage application [Fig. 1(d)]. Interestingly, the ACM started to develop
again immediately after the applied voltage was returned to 0 V [Fig. 1(e)-(g)].
Furthermore, we confirmed that this ON/OFF switching of the self-spreading
behavior could be repeatedly observed, corresponding to the temporal switching
of the applied voltage [Fig. 1(h)-(l)]. The electric double layer, which
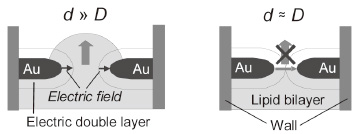
is characterized by the Debye length, plays a crucial role in this behavior.
In a bulk condition, the electric field is shielded by counterions in the
electrolyte solution. In contrast, the sum of the Debye length (D ) from both electrode surfaces (a few nanometers for a 100 mM NaCl solution)
is close to the nanogap width (d ), the electric field can be effectively applied in the nanogap spacing, leading to the electrostatic trapping of lipid molecules. A continuous supply of lipid molecules is vital for the self-spreading. Therefore, the electric field applied to a nanogap acted as a molecule gate for the self-spreading (Fig. 2). The technique described here constitutes the first demonstration of the temporal and spatial control of biomembrane development in a nanometer-scale device and can provide new opportunities for the realization of nanobio devices.
This work was supported by KAKENHI.
[1] Y. Kashimura et al., Jpn. J. Appl. Phys. 47 (2008) 3248.
[2] Y. Kashimura et al., Jpn. J. Appl. Phys. 49 (2010) 04DL15.
[3] Y. Kashimura et al., J. Am. Chem. Soc. 133 (2011) 6118.
 |
 |
|||||
|
|