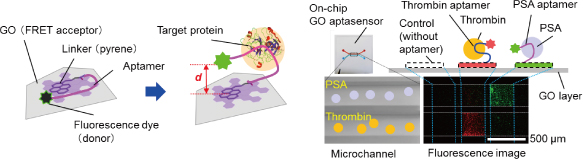
Graphene and its chemical derivatives behave as efficient acceptors for fluorescence resonance energy transfer (FRET) between a graphene surface and dye molecules located close to it. This makes graphene a promising material for realizing a unique type of biomolecular interface for visualizing an invisible biological response to a measurable physical quantity such as fluorescence. We have successfully demonstrated a graphene oxide (GO) aptasensor for selective and highly sensitive protein detection. We realized this sensor by modifying the GO surface with a pyrene-aptamer-dye probe that we developed. The segments work as a linker to the GO surface, a protein recognition part, and a fluorescence detection tag, respectively [Fig. 1 (left)] [1]. The system allows us to perform molecular detection on a solid surface, which is a powerful tool for realizing an on-chip sensor, and especially for forming a multichannel configuration and for micropatterning probes [2, 3]. The on-chip sensor allows us to evaluate the sensor response quantitatively by using one of the channels/patterns as an internal standard.
Aptamers offer many advantages as molecular recognition probes because they are chemically stable and have a wide variety of targets. We confirmed the versatility of the GO aptasensor through the detection of three different proteins, namely, thrombin, prostate specific antigen (PSA), and hemagglutinin, simply by changing the aptamers but with the sensor composition remaining the same. We also demonstrated the simultaneous detection of multiple proteins, thrombin and PSA, on a single chip by using a multichannel linear-array GO aptasensor [Fig. 1 (right)] [3]. Another attractive feature of aptamers is that they can be flexibly designed without loss of activity. We designed a biomolecular probe for highly sensitive protein detection by modifying an aptamer with a DNA spacer. The spacer controls the distance between a fluorescence dye and a quencher, which is crucial for FRET-based sensors. We successfully demonstrated an improvement in the sensitivity of an on-chip GO aptasensor [2]. Our findings will advance the field of on-chip microsensors.