Thermal Instability of Graphene on a Substrate
Graphene is a one-atom-thick thin film consists of the honeycomb lattice of carbon. Graphene is generally considered to be thermally and chemically stable because of its robust sp2 bonding with no dangling bonds. In a practical graphene sample, however, the situation is complex. Graphene often contacts a substrate, metal electrodes, gas molecules in the environment, and residue of PMMA which is commonly used as a protective layer in the transfer process. We show that a practical graphene sample obtained by the commonly used growth and transfer techniques is not stable against heating in a high vacuum [1]. The origin of the heating-induced structural instability is also studied.
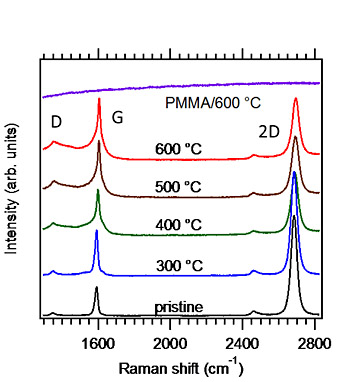
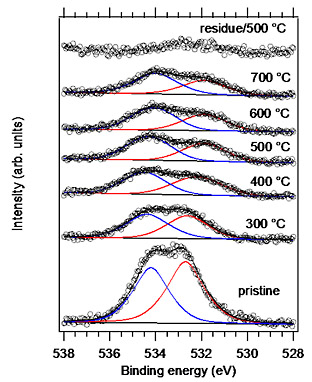
In this study, we only used very common techniques for both CVD growth and transfer. Single-layer graphene was grown on Cu foil by the low-pressure CVD method. A protective PMMA layer was formed on the graphene film on the Cu foil by spin-coating. The PMMA/graphene film was isolated by dissolving the Cu foil in an FeCl3 solution and the film was transferred to a substrate in deionized water. Finally, the PMMA film was removed with acetone. Raman spectra of graphene transferred to a SiO2 substrate before and after heating in a high vacuum are shown in Fig. 1. A broad spectrum appears with heating. The spectral broadenings mean that the Raman selection rule is relaxed due to disorder induced by heating. Figure 2 shows O 1s XPS of a graphene/Au sample before and after heating at several temperatures. The integrated intensity almost stays constant at 400oC and higher temperatures. The binding energy values are close to those of physisorbed H2O and O2 molecules. These results show that the oxygen-containing molecules are inserted between the graphene and the substrate and that the molecules cannot be easily eliminated even in an ultrahigh vacuum and at a high temperature. The structural instability is likely due to defect formation caused by reactions with H2O and O2 molecules underneath graphene.
- [1]
- S. Suzuki, C. M. Orofeo, S. Wang, F. Maeda, M. Takamura, and H. Hibino, J. Phys. Chem. C 117 (2013) 22123.
 |
 |
|
|